- Your cart is empty
- Continue Shopping
Product
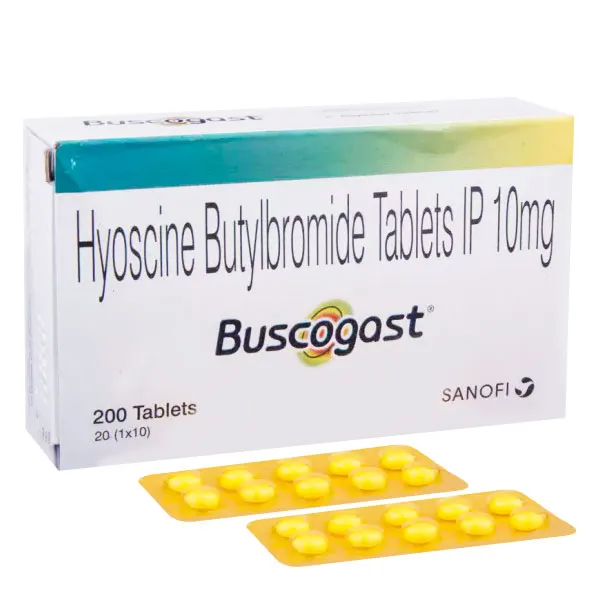
Buscogast (Hyoscine Butylbromide)
Price range: $98.00 through $204.00
Buscogast (Hyoscine Butylbromide) is an antispasmodic medication that relieves abdominal cramps, bloating, and gastrointestinal or urinary discomfort. It works by relaxing smooth muscles, providing targeted relief from spasms. Buscogast treats conditions like irritable bowel syndrome (IBS), indigestion, and period pain. Safe for most adults, it should be used as directed by a healthcare professional. Consult trusted sources like FDA, Mayo Clinic, and Healthline for complete guidance on use, side effects, and interactions before starting treatment.
Buscogast, containing Hyoscine Butylbromide, is an antispasmodic medication designed to relieve abdominal cramps, bloating, and discomfort caused by gastrointestinal and genitourinary spasms. It works by relaxing the smooth muscles in the digestive and urinary tracts, providing targeted relief. Buscogast is commonly used to treat conditions like irritable bowel syndrome (IBS), indigestion, and period pain. Learn more about its medical profile at Mayo Clinic or WebMD.
Key Benefits & Uses
-
Relieves abdominal cramps from gastrointestinal spasms.
-
Alleviates bloating and discomfort caused by trapped gas.
-
Provides relief from indigestion and period pain.
-
Helps manage symptoms of irritable bowel syndrome.
-
Reduces bladder spasms.
For detailed studies, visit PubMed or check treatment guidelines on WHO.
How to Use
-
Adults and children over 6 years: 1–2 tablets 3 times daily.
-
Swallow tablets whole with water; can be taken with or without food.
-
Follow your healthcare professional’s directions for duration.
For official guidance on medication use, see FDA and MedlinePlus.
Important Safety Information
-
Avoid if allergic to hyoscine butylbromide or any component of the tablet.
-
Not recommended for patients with myasthenia gravis, glaucoma, megacolon, or paralytic ileus.
-
Consult a doctor before use during pregnancy or breastfeeding.
-
May interact with other medications; inform your doctor about all current prescriptions.
Learn more about safety from Healthline and Mayo Clinic.
Common Side Effects
-
Dry mouth
-
Constipation
-
Dizziness
-
Blurred vision
-
Difficulty urinating
For research-backed details on side effects, check Drugs.com and WebMD.
Why Choose Buscogast
Buscogast offers fast-acting relief from abdominal cramps and discomfort. Its antispasmodic action targets the root cause of pain, providing effective symptom management. With a low incidence of central nervous system side effects, it is suitable for most adults. Always consult a healthcare professional before use to ensure it is appropriate for your condition. More information is available at WHO and PubMed.
Additional Information
| Strength | 10 mg |
|---|---|
| Size | 60 Tablets, 90 Tablets, 120 Tablets |